
The Industry
Lithium ion batteries are a crucial part of the tech ecosystem: they power most, if not all, phones and computers. For reference, this industry is a $30B+ global market today and is projected to grow to a $100B one within 10 years (Statista, 2018).
Stay tuned for more on this…
How they work
Quick reminder: Atoms, Molecules, Chemicals
All things are made of atoms. Atoms with different numbers of electrons, protons and neutrons make up different elements (i.e. why carbon is different from iron). If we put atoms of two different elements together, they might “react” by exchanging electrons or sharing electrons.
Ions = Atoms that have a charge
Positively charged = Too many protons and not enough electrons.
Negatively charged = Too many electrons.
Neutral = Happy and stable.
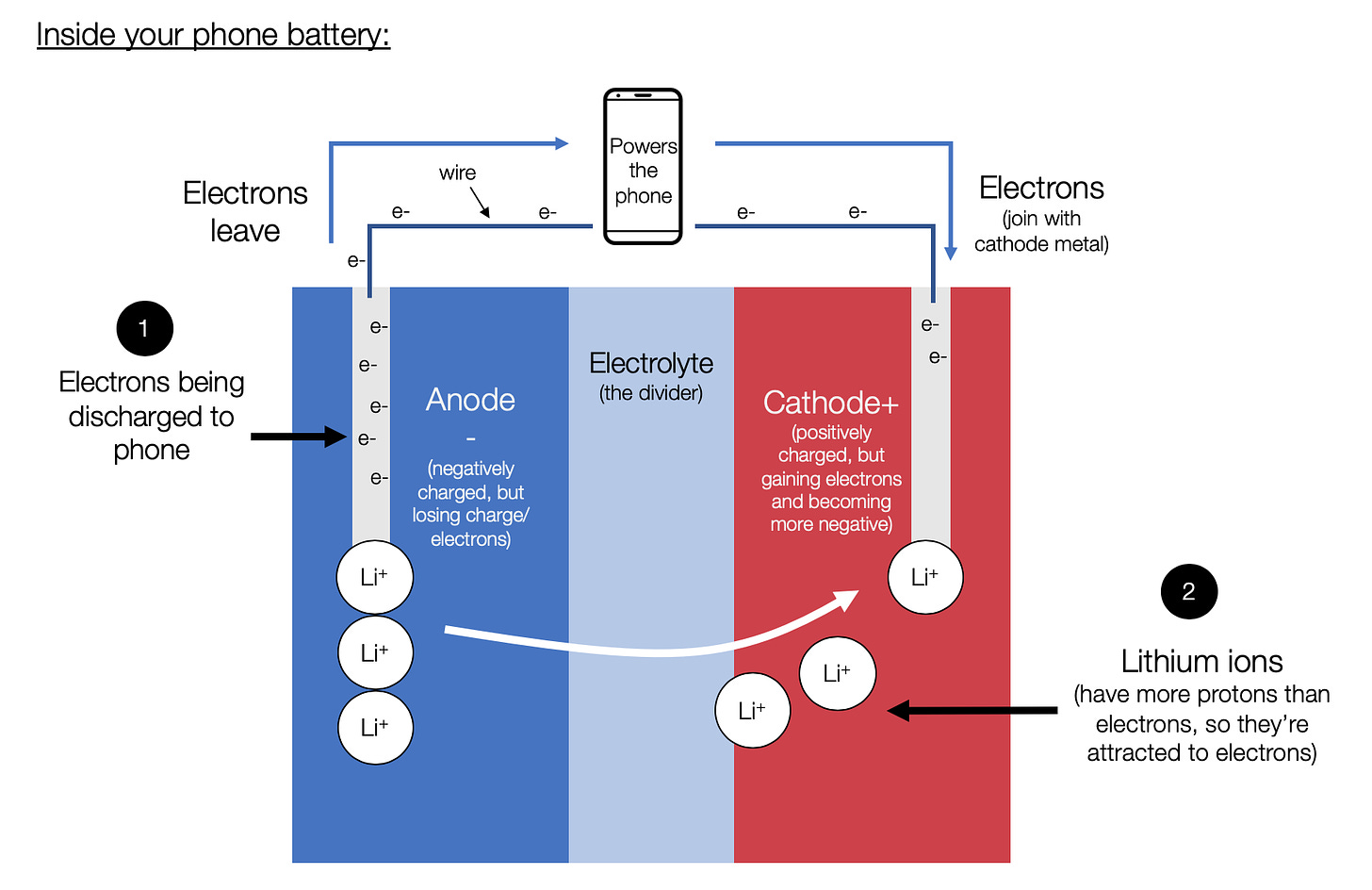
Batteries rely on electrons moving through a wire between two metal plates, which are submerged in lithium ions (Li+). The positively charged plate is the cathode, and the negatively charged one is the anode.
When you’re using your phone:
When the battery is charged and you’re using it, it has an excess of electrons at the anode. These electrons want to move towards a positive charge, so they move through the wire, power the rest of your phone, and then to the positive cathode.
The electrons that begin to collect at the cathode make that part of the battery more negatively charged. The lithium ions that are at the anode are positively charged, so they move across the battery and collect at the cathode too.
When the battery’s dead:
When your phone battery’s all used up, there are no more lithium ions at the anode.

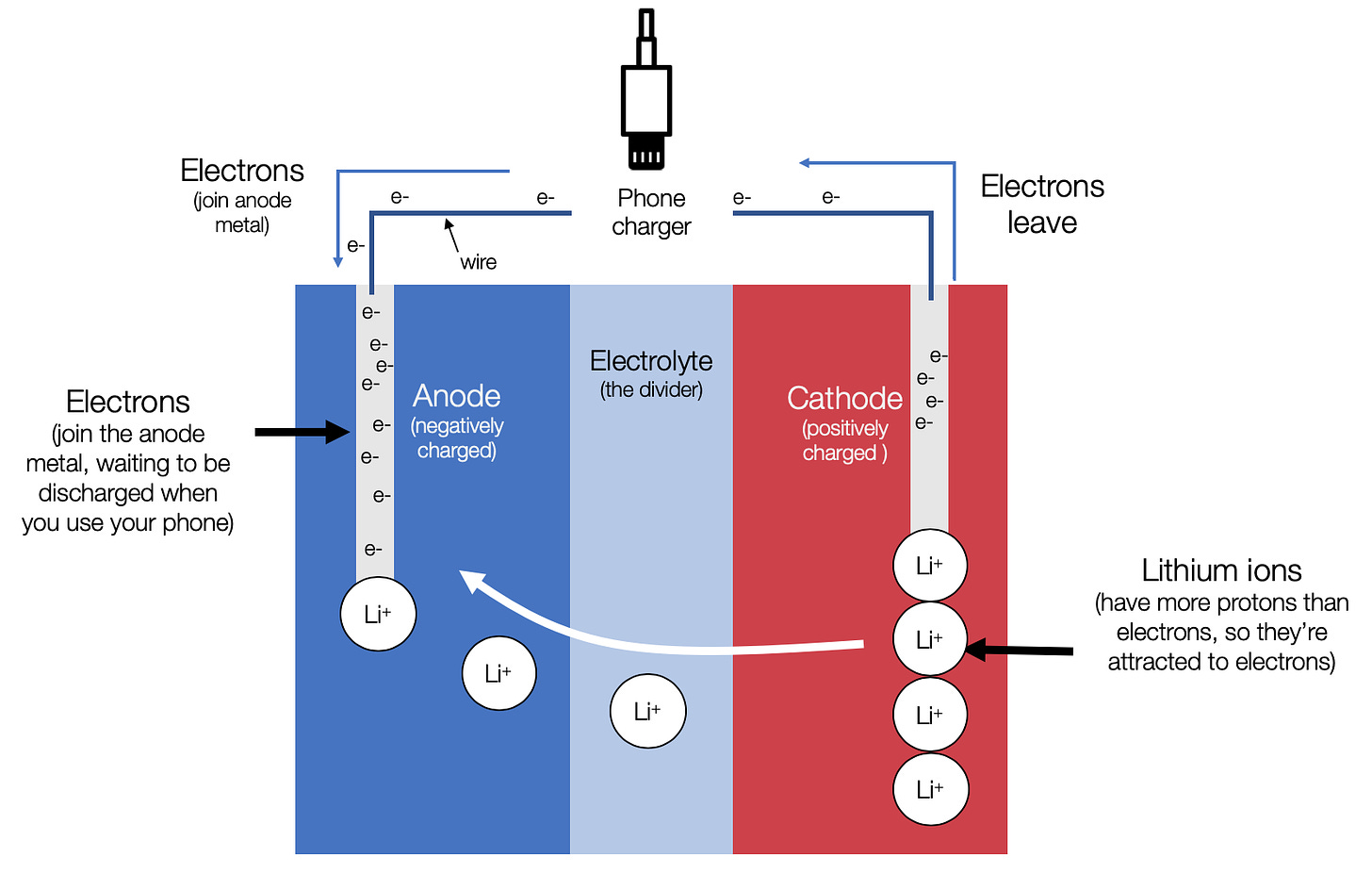
When it’s charging up:
You can pump electricity back into the chemicals and undo the reaction, so you can get them to react over and over again (i.e. a rechargeable battery).
When you plug in your phone, electrons start moving to the battery’s anode, charging it negatively. The positive lithium ions from the cathode start moving back to the anode because they’re attracted to the negative charge at the anode.
Please remember: this is an oversimplification of the the chemical reactions that take place within a battery.
So where does the electricity to charge your battery come from? Keep following us to learn about power generation.


A concise and informative explainer. Plus, the graphics are fabulous!
Clear understanding in simple language. Explain why these batteries get overheated and creates fire